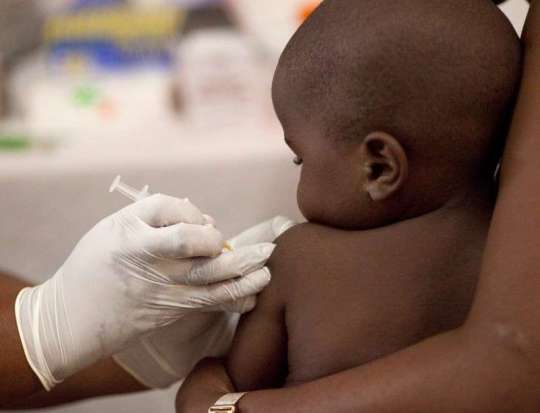
The Ghana Health Service (GHS) has said that measures have been put in place to compensate persons who may be affected adversely after receiving the malaria vaccine.
According to the GHS, a committee has been set up to look into cases of negative health implications to users of the vaccine.
[contextly_sidebar id=”5M7vWziJqW1b5XGdpLMqiQH9VGJsx5rV”]There have been agitations, over the health implications of the new malaria vaccines.
Speaking to Citi News, Director General of GHS, Dr Anthony Nsiah-Asare said the vaccine has been certified by the Food and Drugs Authority, and it’s therefore, safe for use.
He said the “Adverse Event Following Immunization (AEFI) committee” monitors closely the vaccination process and “looks at the causal effect and makes sure that we compensate and manage them when they have any problems.”
“The vaccine has been registered by the Food and Drugs Authority. It is also allowed by the World Health Organisation. It is not true that the drug has not been registered or accepted by the World Health Organisation,” he said.
In the attempt to overcome high child mortality rate resulting from malaria, the Ministry of Health in collaboration with the Ghana Health Service and other partners in April launched a new malaria vaccine as an additional measure to combat the disease.
It is currently being piloted in parts of the country.
As of 2016, Ghana recorded more than ten million suspected cases of malaria with over a thousand deaths.
Speaking at the launch of the new vaccine, Mosquirix, the Director General of the Ghana Health Service, Dr. Nsiah Asare expressed confidence in the outcome of the vaccine.
“As we speak now, we have taken delivery of about 400,000 doses of vaccines. We will continue to monitor the safety of the vaccines. We believe that we are ready to launch the project Godwilling on the 30th of April 2019 at Cape Coast. This vaccination is going to be used to stop malaria from killing our children.”